CDPH Alerts Consumers and Healthcare Providers of Serious Health Risks Related to Counterfeit Botox
What You Need To Know: Counterfeit versions of Botox have been found in multiple states, including California, and are causing hospitalizations and other serious reactions among people who received injections in non-medical, unlicensed settings. Consumers should only get injections of FDA-approved Botox from licensed and trained professionals in healthcare settings.
Sacramento – The California Department of Public Health (CDPH) is alerting consumers and healthcare professionals that counterfeit versions of Botox (botulinum toxin) have been found in multiple states, including California. These counterfeit products have caused hospitalizations and other serious reactions in people who received injections in non-medical, unlicensed settings.
“Counterfeit or incorrectly administered Botox, even in small amounts, can result in serious health problems and even death,” said CDPH Director and State Public Health Officer, Dr. Tomás J. Aragón. “Consumers should only get injections of FDA-approved Botox from licensed and trained professionals in healthcare settings. Botox should never be purchased online or through unlicensed individuals.”
If you have received or are considering Botox injection for medical or cosmetic purposes, confirm with your healthcare professional and/or setting that you are receiving Botox from an authorized source and ask your healthcare professional if they are licensed and trained to administer Botox. If there is any doubt, do not get the injections. In California Botox treatments may be performed by a physician or by a registered nurse or physician assistant under a physician's supervision. More information on cosmetic procedures can be found on the Medical Board of California’s website.
If you have any symptoms of botulism poisoning as described below, contact a healthcare professional or go to the emergency room immediately. Symptoms caused by counterfeit Botox are similar to botulism poisoning and include drooping eyelids, difficulty swallowing, dry mouth, slurred speech, difficulty breathing, fatigue, and generalized weakness.
Consumers should report suspected counterfeit Botox products through the FDA’s website or by calling 800-551-3989. Suspected counterfeit Botox products in California can be reported to CDPH at Consumer Complaints.
Information For Consumers: If you have received or are considering Botox injection for medical or cosmetic purposes, take these precautions:
- Confirm with your healthcare professional and/or setting that you are receiving Botox from an authorized source.
- Ask your healthcare professional if they are licensed and trained to administer Botox.
- If in doubt, do not get the injection.
- Contact a healthcare professional or go to the emergency room immediately if you have any symptoms of botulism poisoning.
Information For Healthcare Professionals: Botulism is considered a medical and public health emergency. If you suspect botulism in a patient, contact your local health department immediately and consider the following:
- Report to your local health department any adverse events following the administration of Botox.
- Check the product for any signs of counterfeiting before administering.
- Federal law requires that all healthcare providers who dispense or administer prescription drugs purchase products only from authorized sources. Purchasing and administering counterfeit products is illegal and puts patients at risk.
- Visit the FDA’s website for information about how to safely purchase prescription drugs for your patients: Know Your Source: Protecting Patients from Unsafe Drugs.
Verifying Authentic Botox: Botox is manufactured by AbbVie, Inc. The authentic product may be identified by the following:
- The product descriptions on the outer cartons are either: “BOTOX® COSMETIC / onabotulinumtoxinA / for Injection” or “OnabotulinumtoxinA / BOTOX® / for injection.”
- The manufacturer identified on the outer carton is either “Allergan Aesthetics / An AbbVie Company” or “abbvie.”
- FDA-approved Botox displays the active ingredient as “OnabotulinumtoxinA” on the outer carton and vial.
Currently, there is no indication that the reported events were linked to AbbVie’s FDA-approved Botox, and the genuine product should be considered safe and effective for its intended and approved uses.
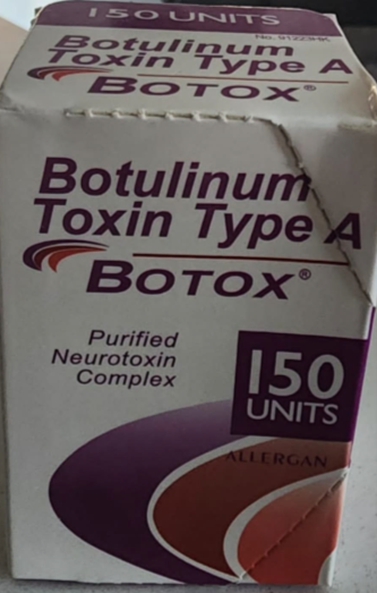
Signs of Counterfeit Versions of Botox: The counterfeit product may be identified by one or more of the following:
- Outer carton/ packaging:
- Displays the active ingredient as “Botulinum Toxin Type A” instead of “OnabotulinumtoxinA”
- Indicates 150-unit doses
- Contain lot number C3709C3
- Includes language that is not English.
- Indicates 150-unit doses
- Contain lot number C3709C3
Examples of Counterfeit Packaging:
| 
|
Examples of Counterfeit Vials: