Injection Safety Information for Healthcare Providers and Public Health Departments
About
Injection safety is a basic expectation in patient care. Injection safety includes safe handling of medications and using each needle and syringe for only one patient one time. Unsafe injection practices have led to transmission of bloodborne pathogens such as hepatitis B and C, HIV, and bacteria.
Injection Safety information for patients and families is also available on the HAI Program’s site.
Impact of Unsafe Injection Practices
 From 2008-2018, 63 outbreaks of healthcare-associated viral hepatitis in the United States were reported to the Centers for Disease Control and Prevention (CDC); 10 of those occurred in California. The California outbreaks resulted in 50 confirmed cases of hepatitis B or C and nearly 4,723 people were advised to get tested for bloodborne pathogens.
From 2008-2018, 63 outbreaks of healthcare-associated viral hepatitis in the United States were reported to the Centers for Disease Control and Prevention (CDC); 10 of those occurred in California. The California outbreaks resulted in 50 confirmed cases of hepatitis B or C and nearly 4,723 people were advised to get tested for bloodborne pathogens.
Healthcare-associated hepatitis linked to unsafe injection practices has occurred in a variety of settings, including nursing homes, assisted living facilities, pain management clinics, outpatient dialysis clinics, and outpatient alternative medicine practices. Common exposures include unsafe injection practices, podiatry in long-term care facilities, drug diversion, and transfusions.
How Does Drug Diversion Lead to Unsafe Injection Practices?
Drug diversion refers to legally prescribed controlled substances obtained and used illicitly (see CDC Drug Diversion Infographic (PDF)). When a healthcare worker with hepatitis C or any other bacterial infection tampers with injectable drugs such as opioids in their workplace, they put patients at risk of acquiring those infections. Many outbreaks related to drug diversion by healthcare workers have been reported to CDC.
Are Injection Safety Breaches Reportable?
Yes. Report breaches and lapses of injection safety and infections suspected of being associated with unsafe practices to your local health department. If you are a licensed facility, you must also report to a CDPH Licensing and Certification district office.
How Do I Follow Safe Injection Practices?
Follow CDC's Core Infection Prevention and Control Practices for Safe Healthcare Delivery in All Settings (PDF) and CDC's One and Only Campaign for safe injection and medication practice recommendations.
- Perform hand hygiene and use aseptic technique when preparing and administering medications
- Before entry, always disinfect the tops of medication vials with 70% alcohol; allow tops to dry before inserting a needle or device into the vial
- Draw up medications in a clean medication area; the designated medication area should not be near areas where contaminated items are placed
- Always use needles and syringes for one patient only, including manufactured prefilled syringes and cartridge devices such as insulin pens
- Enter medication containers with a new syringe and a new needle even when obtaining additional doses for the same patient
- Ensure single-dose or single-use vials, ampules, bags or bottles of parenteral solution, fluid infusion and administration sets (for example, intravenous tubing) are used for one patient only!
- Use single-dose medication vials whenever possible
- Read the label on medication vials carefully to determine if it’s for single use; if the vial says single-dose, throw it away after it has been accessed
- Do not save single-dose medications for future use!
- Discard unused single-dose medications when expired
- Limit the use of multi-dose medication vials whenever possible
- A multi-dose vial is recognized by its FDA-approved label
- Dedicate multi-dose vials to a single patient whenever possible
- If multi-dose vials must be used for more than one patient, restrict the medication vials to a centralized medication area and never take them into the patient treatment area (such as operating room, patient room/cubicle)
- Discard multi-dose vials when the beyond-use date has been reached
- Any time the sterility of the vial is in question, throw it out
- Date the multi-dose vial when first opened; discard within 28 days, unless the manufacturer recommends a shorter expiration period
- Wear a facemask when placing a catheter or injecting material into the epidural or subdural space (such as during myelogram, epidural, or spinal anesthesia)
Vaccination Safety Tips
- Use manufactured single-dose, pre-filled syringes if possible
- If multi-dose vials of vaccine must be used, ensure
- All safety precautions are taken to avoid contamination of multi-dose vials
- Correct dose is administered
- Prepare vaccine in a clean, designated space away from contaminated or dirty areas
- Do not pre-draw vaccines before patients arrive at clinic
- For more information refer to CDC’s Vaccine Storage and Handling Toolkit (PDF)
Some Special Considerations for Diabetic Patients
Diabetic patients use needles frequently in the care and management of their disease
How can I Educate My Staff about Injection Safety?
How Can I Assess my Facility’s Injection Safety Practices?
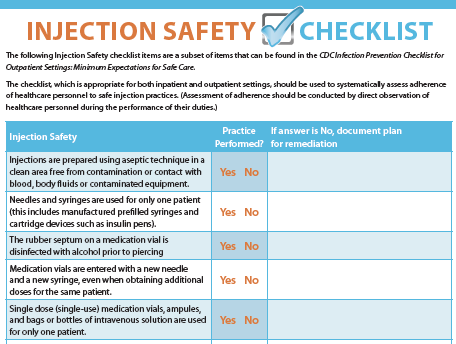 Use a Checklist
Use a Checklist
Use an injection safety checklist as a training resource and to audit injection safety practices of healthcare workers.
CDC Injection Safety Checklist (PDF)
Monitor  Injection Practices
Injection Practices
Use a standardized adherence monitoring tool to measure adherence to safe injection practices in any location where medication is prepared or provided to patients. Adherence monitoring will help to identify areas for potential harm to patients or healthcare workers.
CDPH Injection Safety Adherence Monitoring Tool (PDF)
 Observe Hand Hygiene Adherence
Observe Hand Hygiene Adherence
Use a standardized adherence monitoring tool to identify gaps and opportunities for improvement. Monitoring may be performed in any type of patient care location.
CDPH Hand Hygiene Adherence Monitoring Tool (PDF)
 Evaluate Blood Glucose Monitoring
Evaluate Blood Glucose Monitoring
Observe blood glucose monitoring practices and identify gaps and opportunities for improvement. Monitoring may be performed in any type of patient care location where blood glucose is monitored.
CDPH Blood Glucose Meter Adherence Monitoring Tool (PDF)
Assess Infection Control Practices
CDC developed assessment tools to assess overall infection control programs and practices for acute care (including hospitals and long-term acute care hospitals), outpatients, long-term care, and hemodialysis settings.
Evaluate Your Sharps Injection Prevention Program
Ensure healthcare workers are doing all they can to prevent needlesticks and other sharps-related injuries.
Public Health Role in Injection Safety
Public health partners should be aware of injection safety and be familiar with resources for investigating healthcare-associated cases of acute hepatitis. Below are general recommendations:
- CDPH developed a quicksheet to guide local health departments through the steps of investigating healthcare-associated hepatitis B and C in healthcare facilities
- CDPH developed a presentation about investigating cases of acute viral hepatitis with healthcare exposures
- Report acute hepatitis B or C cases
- CDC’s Viral Hepatitis and Healthcare-Associated Outbreaks webpage contains helpful resources including a healthcare investigation checklist, healthcare investigation guide, healthcare investigation letter example, healthcare notification and testing toolkit
For information on how to develop documents for patient notification, plans for media and communication strategies, establish communication resources, and best practices in conducting patient notifications, visit CDC’s Patient Notification Toolkit
CDC developed a toolkit tailored to state and local health departments with information on impacts related to unsafe injection practices, guide for building a working group, guide to developing partnerships, tips for media/press relations, and other key resources
The Council of State and Territorial Epidemiologists (CSTE) released a toolkit for state and local health departments on drug diversion planning and response. The toolkit highlights response to drug diversion issues, offers resources and emphasize collaborations among public health organizations representing regulatory affairs such as law enforcement, healthcare facility licensing, certification and accreditation
Additional Resources